Design and sizing of passive systems for treatment of circum-neutral ferruginous mine water
2 BASE TECHNOLOGIES GmbH
O 1.5 in The Skin of the Earth and below: Soil and Water
11.10.2018, 10:10-10:25, H36, NW III
Contamination of water resources by mining activities is one of the most complex and costly environmental problems worldwide. Most prominent is the occurrence of Acid Mine Drainage as a result of sulphide mineral oxidation, releasing ferrous iron, acidity, sulphate and associated metals into aqueous environments. Subsequent aeration and neutralisation leads to precipitation of ferric hydroxides, resulting in impaired usability of water resources and/or complete destruction of aquatic ecosystems. Ferruginous mine water can be treated in passive systems by using and enhancing natural processes. However, the current approach to conceptualising and sizing of passive systems for iron removal from circum-neutral mine water doesn’t reflect the kinetic background of respective biogeochemical and physical processes.
A pilot plant was installed for passive treatment of ferruginous seepage water from a former lignite open-pit. The innovative system consists of three identical lines with multiple successive treatment stages each:
- Preliminary treatment in sedimentation ponds,
- fine treatment in wetland-cells and
- purification in sediment filters.
System monitoring includes fixed sensors for continuous measuring of hydrochemical parameters (pH, conductivity, O2, turbidity) and periodic sampling over the course of 3 years.
Preliminary results show very good iron removal rates, successively reducing concentrations from ±10mg/l in the influent to <1mg/l in the effluent. High-resolution monitoring reveals a strong impact of environmental factors (season, day/night, weather) on hydrochemistry that might affect treatment efficiency. Also, a strong relationship between turbidity and iron is observed.
In the month to come, hydraulic variation as well as additional measurements and experiments will be used to investigate kinetics and relations of the decisive iron removal reactions/processes to develop a model concept and ultimately a new sizing approach for passive systems.
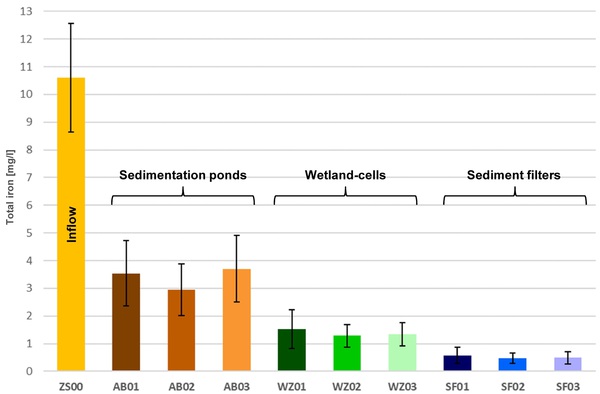
Successive iron removal by multiple treatment components
Keywords: biogeochemistry, mine water, passive treatment, iron, wetland
Export as iCal:



